InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Peptide CDMO 2.0 Market”-, By Product Type / Modality (Linear peptides, Cyclic peptides, Stapled peptides, Peptide–drug conjugates (PDCs), Peptide–oligonucleotide conjugates, GLP-1 and related long-acting analogues, Oral peptide formulations), By Scale of Operation (Preclinical, Clinical (Phase I–III), Commercial), By Business Model (Tech-Enabled CDMOs, Niche-Focused CDMOs, End-to-End Integrated CDMOs, Sustainability-Driven CDMOs), By Technology Platform (Solid Phase Peptide Synthesis (SPPS), Liquid Phase Peptide Synthesis (LPPS), Hybrid SPPS–LPPS, Enzymatic/biocatalytic synthesis, AI-assisted synthesis optimization), By Therapeutic Area (Oncology, Metabolic disorders, Infectious diseases, Rare & genetic disorders, Cardiovascular, Neurology), By End User (Large pharmaceutical companies, Emerging biotechs, Academic & research institutions), and Global Forecasts, 2025-2034 And Segment Revenue and Forecast To 2034.”
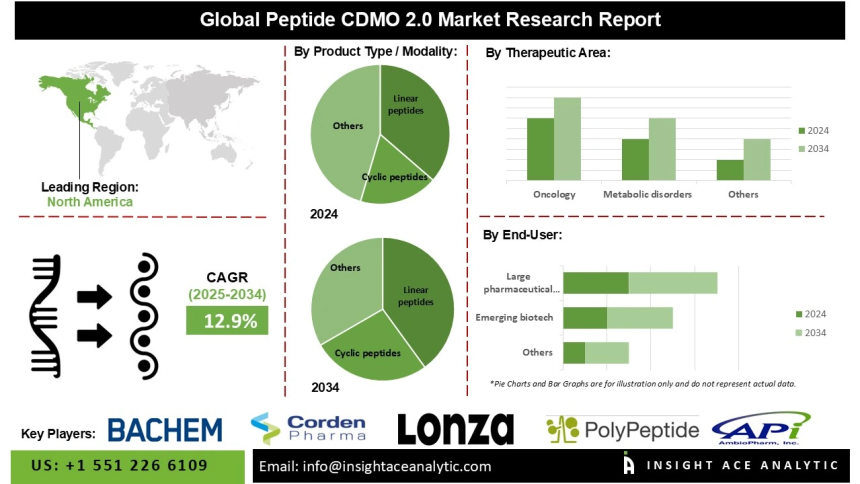
Global Peptide CDMO 2.0 Market Size is predicted to grow at a 12.9% CAGR during the forecast period for 2025-2034.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3242
Peptide CDMOs 2.0 are placing greater emphasis on flexible and scalable manufacturing frameworks, leveraging modular facility designs and multipurpose production systems that enable efficient alignment with evolving regulatory requirements and the diverse specifications of peptide-based therapeutics. The capability to scale production effectively—ranging from managing variable demand cycles to supporting large-volume manufacturing of high-demand compounds such as GLP-1 receptor agonists—is essential for producing peptides with complex structures and diverse sequences.
These organizations support the supply of research-grade peptides, the development of peptide libraries for high-throughput screening, the production of chemically modified peptides (including stapled, cyclic, and PEGylated variants) to improve stability and bioactivity, and the facilitation of target validation studies to evaluate protein–protein interactions and receptor binding. Collectively, these services position peptide CDMOs as key contributors to both early-stage drug discovery and commercial-scale manufacturing.
The evolution toward Peptide CDMO 2.0 has significantly transformed traditional manufacturing approaches through the adoption of automation, artificial intelligence-driven process optimization, continuous manufacturing technologies, and advanced data analytics. These innovations have streamlined development timelines, enhanced production yields, and improved overall cost efficiency.
The implementation of continuous-flow systems, alongside advanced synthesis methods such as solid-phase and liquid-phase peptide synthesis, has strengthened scalability and reproducibility, enabling smooth transitions from clinical development to full-scale commercial production. In addition, these capabilities support the advancement of complex peptide modalities, including cyclic peptides and peptide–drug conjugates, while contributing to reduced time-to-market.
Read Comprehensive Report Overview: https://www.insightaceanalytic.com/report/peptide-cdmo-2-0-market/3242
The growing demand for peptide-based therapeutics across therapeutic areas such as oncology, metabolic disorders, cardiovascular diseases, and infectious diseases is driving substantial expansion within the peptide CDMO market. Increased reliance on outsourcing by both established pharmaceutical companies and emerging biotechnology firms, along with broader regulatory acceptance of peptide therapeutics and rising investment in biologics research, is further accelerating market growth. CDMOs equipped with advanced infrastructure, AI-enabled technologies, and integrated end-to-end manufacturing capabilities are well positioned to facilitate the commercialization of next-generation peptide therapies, thereby playing a pivotal role in the advancement of precision medicine.
List of Prominent Players in the Peptide CDMO 2.0 Market:
- Lonza Group AG
- CordenPharma
- Bachem Holding AG
- AmbioPharm
- PolyPeptide Group
- Evonik Health Care
- WuXi AppTec / WuXi TIDES
- Thermo Fisher Scientific (Patheon)
- Olon S.p.A.
- NOF Corporation
- Curapath
- eTheRNA Manufacturing
- Helix Biotech
- Phosphorex
- Creative Peptides
- Peptron Inc.
- Pepscan
- CSBio
- Neuland Laboratories
- Asymchem
- Sai Life Sciences
- AmbioPharm Shanghai
- Hybio Pharmaceutical
Market Dynamics
Drivers:
The rising global burden of chronic diseases—including obesity, cancer, and cardiovascular disorders—is fueling strong demand for peptide- and oligonucleotide-based therapeutics. These treatment modalities enable highly targeted and individualized therapies, making them particularly suitable for managing complex, long-term conditions. As the incidence of chronic diseases continues to increase, pharmaceutical companies are increasingly partnering with Contract Development and Manufacturing Organizations (CDMOs) to advance the development of innovative peptide- and oligonucleotide-based solutions. In parallel, the broader shift toward precision medicine is further reinforcing the demand for these advanced therapeutic approaches.
The expanding use of peptides across a wide range of therapeutic areas—including oncology, metabolic diseases such as diabetes and obesity, infectious diseases, neurology, and rare disorders—is a key growth driver. High-demand therapies, including GLP-1 receptor agonists such as Semaglutide and Liraglutide, highlight the need for scalable and high-capacity manufacturing infrastructure. In addition, chemically modified peptides—such as cyclic, stapled, PEGylated, and lipidated variants that offer enhanced stability, bioavailability, and efficacy—are gaining widespread adoption. The development of Peptide–Drug Conjugates (PDCs) and Peptide–Oligonucleotide Conjugates further underscores the industry’s transition toward precision and personalized medicine, emphasizing the importance of advanced CDMO capabilities.
Challenges:
Peptide manufacturing remains highly complex and capital-intensive, creating barriers for startups and smaller organizations. Regulatory requirements governing peptide therapeutics are stringent, often necessitating compliance with multiple standards enforced by authorities such as the U.S. Food and Drug Administration (FDA). Extended approval timelines and significant compliance-related expenditures can restrict market entry and slow commercialization for some companies.
Regional Trends:
North America is expected to maintain the leading position in the market over the forecast period, supported by significant investment in peptide therapeutics and a strong foundation in biopharmaceutical research and development. CDMOs in the region are well positioned to provide specialized synthesis and development services, including the production of high-purity linear and cyclic peptides, aligning with the growing emphasis on biologics and personalized medicine.
The Asia-Pacific region is projected to experience the fastest growth, driven by favorable cost advantages, rapid expansion of pharmaceutical infrastructure, and increasing demand for peptide-based therapeutics. CDMOs in the region are increasingly integrating advanced technologies such as AI-driven process optimization, continuous-flow manufacturing, and automated solid-phase peptide synthesis (SPPS) to improve efficiency and support innovation. For instance, in January 2024, WuXi TIDES expanded its manufacturing capacity for linear and cyclic peptides by establishing two advanced facilities in Changzhou and Taixing, China, incorporating automated SPPS systems and digitalized production processes to support its expanding therapeutic pipeline.
Add our site to Google Preferred Sources for quality content: https://google.com/preferences/source?q=insightaceanalytic.com
Recent Developments:
- In January 2024, WuXi AppTec launched two new peptide manufacturing plants, one in Changzhou and another at their new Taixing API site in China, tripling their overall peptide synthesis capacity and increasing total Solid-Phase Peptide Synthesis (SPPS) reactor volume to 32,000 liters. These advanced facilities use digital operations and automated solvent delivery systems to improve production efficiency, consistency, and scalability.
- In May 2023, PolyPeptide and Numaferm signed a Preferred Partner Collaboration Agreement for peptide development and production, utilizing Numaferm’s biochemical production platform and sustainable peptide manufacturing expertise, as well as PolyPeptide’s cGMP manufacturing capabilities, regulatory expertise, and market access. The company specializes in the development and production of peptides and proteins. The parties have committed to maintaining the confidentiality of the agreement’s specifics.
Global Peptide CDMO 2.0 Market- By Product Type / Modality
- Linear peptides
- Cyclic peptides
- Stapled peptides
- Peptide–drug conjugates (PDCs)
- Peptide–oligonucleotide conjugates
- GLP-1 and related long-acting analogues
- Oral peptide formulations
Global Peptide CDMO 2.0 Market – By Scale of Operation
- Preclinical
- Clinical (Phase I–III)
- Commercial
Global Peptide CDMO 2.0 Market – By Business Model
- Tech-Enabled CDMOs (automation, AI, data integration)
- Niche-Focused CDMOs (rare diseases, complex peptides)
- End-to-End Integrated CDMOs
- Sustainability-Driven CDMOs
Global Peptide CDMO 2.0 Market- By Technology Platform
- Solid Phase Peptide Synthesis (SPPS)
- Liquid Phase Peptide Synthesis (LPPS)
- Hybrid SPPS–LPPS
- Enzymatic/biocatalytic synthesis
- AI-assisted synthesis optimization
Global Peptide CDMO 2.0 Market – By Therapeutic Area
- Oncology
- Metabolic disorders (incl. obesity/diabetes)
- Infectious diseases
- Rare & genetic disorders
- Cardiovascular
- Neurology
Global Peptide CDMO 2.0 Market – By End User
- Large pharmaceutical companies
- Emerging biotech
- Academic & research institutions
Global Peptide CDMO 2.0 Market – By Region
North America-
- The US
- Canada
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Why should buy this report:
- To receive a comprehensive analysis of the prospects for the global Peptide CDMO 2.0 Market. To receive an industry overview and future trends of the global Peptide CDMO 2.0 Market
- To analyze the Peptide CDMO 2.0 Market drivers and challenges
- To get information on the Peptide CDMO 2.0 Market. size value (US$ Mn) forecast till 2034
- Major Investments, Mergers & Acquisitions in the Cloud-Based and AI-Driven Eye Tracking Systems industry
Customize this Study according to your Requirements @ https://www.insightaceanalytic.com/customization/3242
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com